Research
Research Background
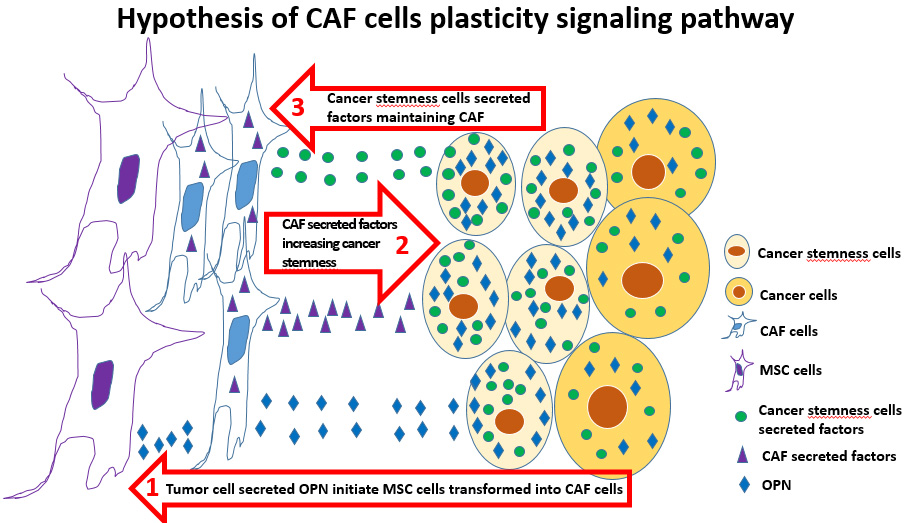
Cancer cells interact with the tumor microenvironment to create a milieu that enhances local and distant tumor potentiation. Tumor growth and metastasis is highly dependent on complex dynamic interactions between cancer cells and tumor stroma, mediated by direct cell–cell contact and secreted growth factors and cytokines. Cancer-associated fibroblasts (CAFs), a tumor stromal element, are involved in tumor growth potentiation, extracellular matrix degradation, tumor cell motility, inhibition of host antitumor response, promotion of angiogenesis, and metastasis. CAF origin is multifactorial and is derived from local fibroblasts, bone marrow–mesenchymal stem cells (MSCs), pericytes, and tumor cells. It is generally accepted that α-smooth muscle actin (SMA), tenascin-C (TEN), and vimentin (VIM) describe the CAF phenotype. In addition, transforming growth factor-β (TGF-β) is critical for CAF activation and elaboration of a protumorigenic microenvironment. Signaling by TGF-β regulates tumor initiation, progression, and metastasis through tumor cell–autonomous and host–tumor interactions. Osteopontin (OPN), a phosphoprotein secreted by malignant cells and tumor stromal cells, is a key mediator of tumor cell migration and metastasis and a marker of breast cancer progression and metastasis. Recent findings suggest that tumor-derived OPN instigates bone marrow–derived MSC trafficking to the tumor microenvironment, which is characterized by the outgrowth of a desmoplastic stroma rich in CAFs that promotes cancer growth and metastasis. Within the context of OPN-mediated CAF activation and tumor-microenvironment crosstalk, cancer stem cell (CSC) marker expression (Oct4, Nanog, and Sox2) and metastatic behavior of human breast cancer are enhanced.
Current Study
Our on-going experiments indicate that the cancer cells secret OPN to initiate mesenchymal stem cells transformation into cancer-associated fibroblast cells (CAFs), which would promote the cancer stemness, metastasis and recurrence, and chemotherapy resistance. However, we found OPN itself fails to maintain cancer-associated fibroblast cell transformation. Currently, we are using state of the art techniques such as RNAseq, Crispr screening, xenograft mice cancer models, and patient derived xenograft (PDX) models to characterize the pathways by which CAF promote cancer stemness which feedback to maintain the CAF phenotype.
Long Term Goal
We will develop small molecule drugs such as aptamers to block CAF transformation and cancer stemness in the tumor microenvironment.